Ozone treatment produces water of such superior quality that it cannot be matched with any other type of chemical treatment. Ozone can be used as a disinfectant, decolorisation, deodorisation, detoxifier, precipitant, coagulant and as taste remover. Because of this, it can reduce other chemical loading in treatment facilities.
Drinking water is usually produced from natural sources like groundwater or lakes and rivers (surface waters). Surface water is contaminated with a varying degree of naturally-occurring dissolved organics and metals like sulphides, nitrites, arsenic, synthetic chemicals and micro organisms. It usually has to undergo more purification steps than groundwater to become suitable for drinking.
The numbers of purification steps taken are dependent on the quality of water that enters the purification plant. For surface water treatment, the water is usually stored in a reservoir for several days, in order to improve clarity and taste by allowing more oxygen from the air to dissolve in it and allowing suspended matter to settle down. The water is then pumped to a purification plant through pipelines, where it is treated, so that is will meet government standards.
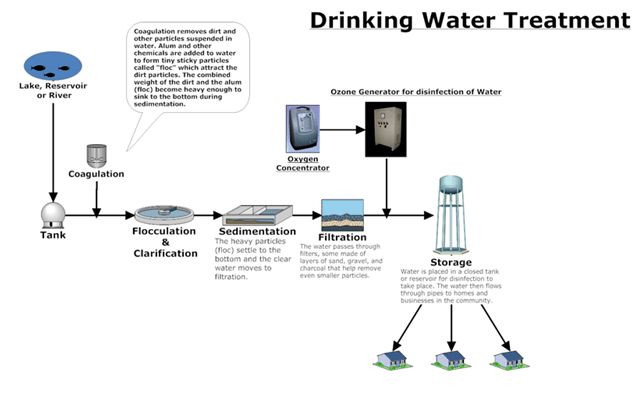
Physical purification is done using screens and filters such as sand filters, RO filters, etc. Chemical purification is carried out using chelating, oxidising, reducing agents (as per needs), coagulants and ion exchange. Disinfection is achieved by addition of chlorine, ozone, hydrogen peroxide, ultra violet rays. Biological water purification is performed to lower the organic load of dissolved organic compounds. Microorganisms, mainly bacteria, do the decomposition of these compounds. There are two main categories of biological treatment: aerobic treatment and anaerobic treatment. The Biological Oxygen Demand (BOD) defines the organic load.
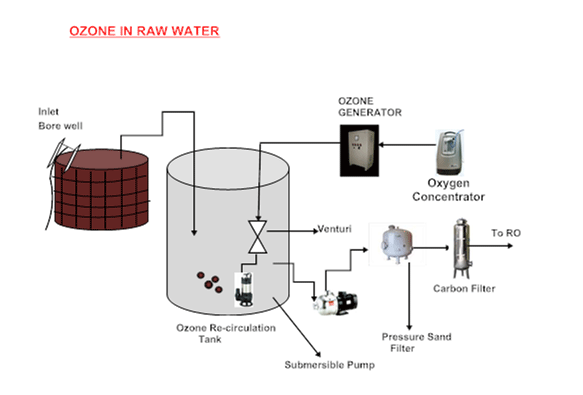
Ozone can be added at several points throughout the treatment system such as during pre-oxidation, and before a sand filter or an active carbon filter (GAC). In water treatment applications with ozone which require ozone gas concentrations between 7 and 15 percent, oxygen is used as feed gas. Also, ozone dosages of up to 100 ppm are common.
Ozone is the most powerful and effective broad spectrum microbiological disinfecting agent available. Ozone causes oxidation and lysis of bacterial cell walls. Inactivation of Giardia cyst requires Destruction of bacteria, viruses, spores, giardia and cryptosporidium with brief contact periods. Ozone Ct value for a 3 log giardia cyst inactivation is 1/100th that of chlorine and 1/1000th that of chloramine. For the treatment of raw water, 1 ppm free chlorine has Ct value of 75 whereas 1 ppm ozone has Ct value of 0.95. Viruses are quickly neutralised by ozone. For cryptosporidia, ozone is the recommended disinfectant.
Oxidation of metals such as iron and manganese by ozone is quick and efficient over a wide pH range. Their insoluble oxides can be easily removed. Nitrite ions can be oxidises with ozone very rapidly, forming nitrate ions which are stable and soluble.
Hydrogen sulphide is responsible for a foul odour (rotten egg) in water. It can be quickly oxidises by ozone in various stages, the last stage being the sulphate ion. sulphate are innocuous and, in many cases, can be filtered. Another important oxidation with ozone is for arsenic. However, in such a case it is important that the raw water contains magnesium or calcium cations. In the presence of these ions, ozone will produce insoluble magnesium and/or calcium ortho-arsenates which will precipitate.
Advanced Oxidation Processes (AOPs) use ozone with UV and hydrogen peroxide and produces OH or hydroxyl radicals, the most powerful oxidants. Advanced Oxidation processes are very fast and are most often used to remove COD or specific organic compounds from water. AOP does not create sludge unlike physical, chemical or biological processes. Non selective pathway allows for the treatment of multiple organics at once.
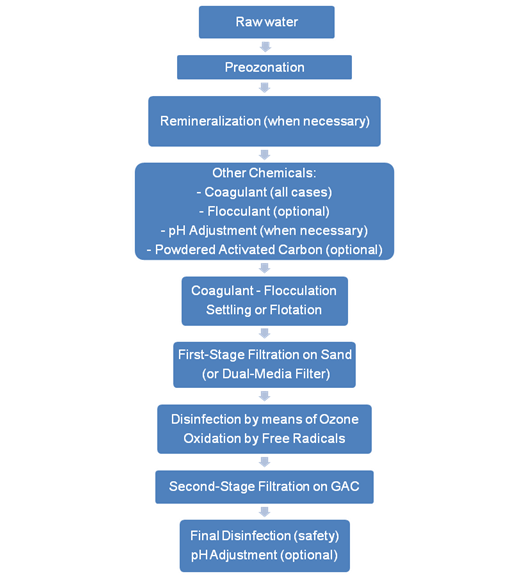
In general, ozonation is accompanied by microflocculation. This enhanced microflocculation effect of ozone can increase filtration rate, decrease the size of filtration beds and lower chemical consumption. Ozone lowers turbidity, lowers particle counts in the filtered water and increases filter runs. Ozone lowers the dose of coagulant, in the range of 20 to 50% when used in pre-ozonation applications. Ozone has a positive effect on COD removal. It breaks down refractory compounds with large and long chain organic molecules into smaller building blocks and making them biodegradable and less dangerous. They are more readily recycled or reused in the environment or removed in filtration process.
Ozone production on site, via normal corona discharge, eliminates the problems of transporting, storing and handling of hazardous and dangerous materials. Oxygen required to make ozone is obtained from air using our oxygen concentrator machine. When ozone is utilised, the chlorine need is greatly reduced, as ozone can totally replace chlorine, chloramine or chlorine dioxide in the pre-oxidation and main oxidation stages. Chlorine is needed only to provide a residual in the distribution system, at 0.2 mg Cl2/L. This minimises the potential for producing carcinogenic disinfection by-products (DBP) such as THMs and helps protect your health. Ozone is of particular value in water treatment systems because of its ability to disinfect without adding other chemicals which may later need to be removed.
Today new ozone drinking water works are lower in investment as well as in the running costs. For this very reason, many cities around the world, such as Los Angeles, Barcelona, Singapore, Paris and Zurich are using ozone for their municipal drinking water plants.
Recent Comments